 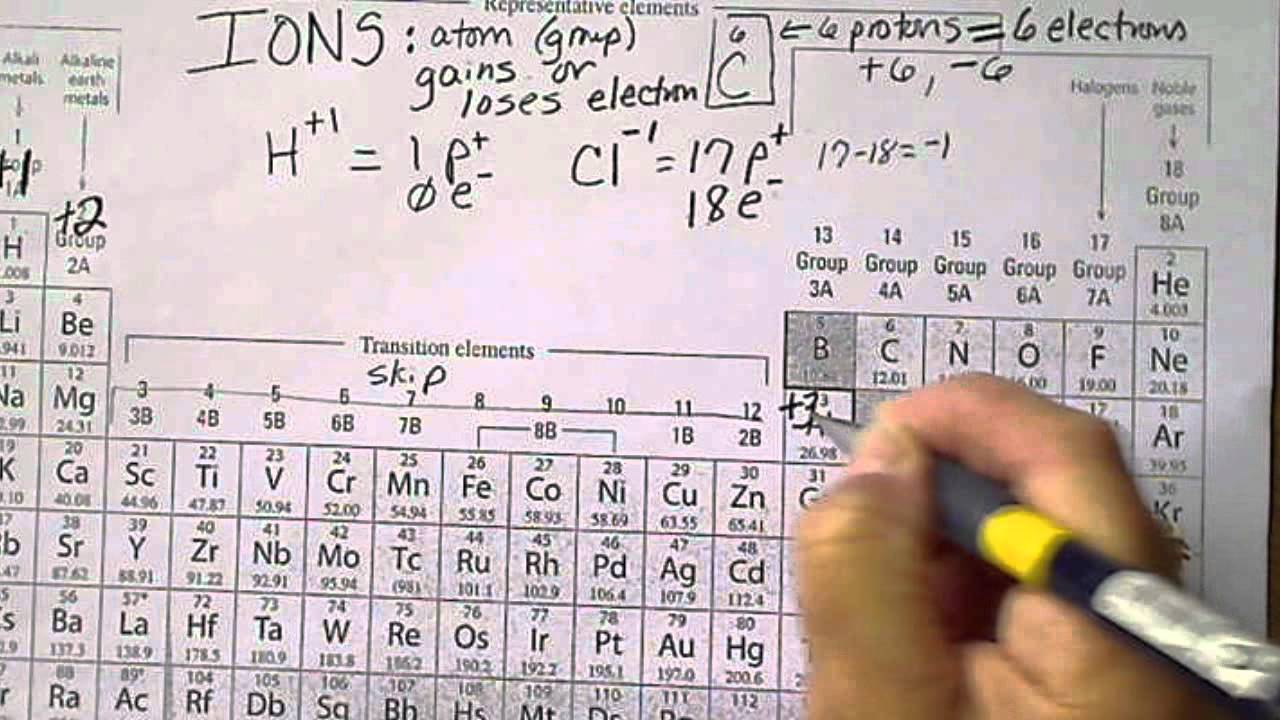
This will be covered in more detail in the next module on nomenclature.Īs you encounter monoatomic and polyatomic ions in different compounds and solutions, it is important to think about the differences in these ions. 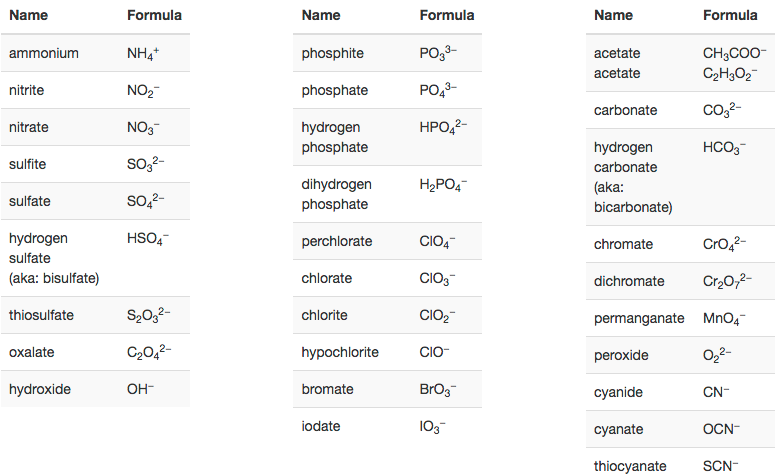
Unfortunately, the number of oxygen atoms corresponding to a given suffix or prefix is not consistent for example, nitrate is NO 3 − while sulfate is SO 4 2−. For example, perchlorate is ClO 4 −, chlorate is ClO 3 −, chlorite is ClO 2 − and hypochlorite is ClO −. Per- (short for “hyper”) and hypo- (meaning “under”) are prefixes meaning more oxygen atoms than -ate and fewer oxygen atoms than -ite, respectively. Note that there is a system for naming some polyatomic ions -ate and -ite are suffixes designating polyatomic ions containing more or fewer oxygen atoms. Oxyanions are polyatomic ions that contain one or more oxygen atoms. Some of the more important polyatomic ions are listed in Table 6.2a. These ions, which act as discrete units, are electrically charged molecules (a group of bonded atoms with an overall charge). The ions that we have discussed so far are called monatomic ions, that is, they are ions formed from only one atom. Write the symbol for each ion and name them. Predict which forms an anion, which forms a cation, and the charges of each ion. The symbol for the ion is N 3−, and it is called a nitride ion.Īluminum and carbon react to form an ionic compound. Thus, a nitrogen atom will form an anion with three more electrons than protons and a charge of 3−. A nitrogen atom must gain three electrons to have the same number of electrons as an atom of the following noble gas, neon. Nitrogen’s position in the periodic table (group 15) reveals that it is a nonmetal. The symbol for the ion is Mg 2+, and it is called a magnesium ion. Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. A magnesium atom must lose two electrons to have the same number electrons as an atom of the previous noble gas, neon. Magnesium’s position in the periodic table (group 2) tells us that it is a metal. Magnesium and nitrogen react to form an ionic compound. Watch Conductivity Molten Salt Video (51 seconds) (credit: modification of work by Mark Blaser and Matt Evans in Chemistry (OpenStax), CC BY 4.0). Figure 6.2a: Sodium chloride melts at 801 ☌ and conducts electricity when molten. When molten, however, it can conduct electricity because its ions are able to move freely through the liquid (Figure 6.2a). (As a comparison, the molecular compound water melts at 0 ☌ and boils at 100 ☌.) In solid form, an ionic compound is not electrically conductive because its ions are unable to flow (“electricity” is the flow of charged particles). 
For example, sodium chloride melts at 801 ☌ and boils at 1413 ☌. Ionic compounds are solids that typically melt at high temperatures and boil at even higher temperatures. You can often recognize ionic compounds because of their properties. However, it is not always true (for example, aluminum chloride, AlCl 3, is not ionic). This guideline works well for predicting ionic compound formation for most of the compounds typically encountered in an introductory chemistry course. The periodic table can help us recognize many of the compounds that are ionic: When a metal is combined with one or more nonmetals, the compound is usually ionic. Similarly, each calcium atom (group 2) can give up two electrons and transfer one to each of two chlorine atoms to form CaCl 2, which is composed of Ca 2+ and Cl − ions in the ratio of one Ca 2+ ion to two Cl − ions.Ī compound that contains ions and is held together by ionic bonds is called an ionic compound. For example, when each sodium atom in a sample of sodium metal (group 1) gives up one electron to form a sodium cation, Na +, and each chlorine atom in a sample of chlorine gas (group 17) accepts one electron to form a chloride anion, Cl −, the resulting compound, NaCl, is composed of sodium ions and chloride ions in the ratio of one Na + ion for each Cl − ion. The compound formed by this transfer is stabilized by the electrostatic attractions (ionic bonds) between the ions of opposite charge present in the compound. When an element composed of atoms that readily lose electrons (a metal) reacts with an element composed of atoms that readily gain electrons (a nonmetal), a transfer of electrons usually occurs, producing ions. Determine formulas for simple ionic compounds.Predict the type of compound formed from elements based on their location within the periodic table.By the end of this section, you will be able to: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
